-

Teoxane to Launch First APAC Regional Academy in Bangkok
New hub strengthens Teoxane's commitment to scientific education and clinical partnerships across Asia-Pacific GENEVA, Jan. 14, 2026 -- Teoxane opens its first APAC Regional Academy in Bangkok, Thailand this January, expanding its global medical-education network. Located on Level 12 of One Bangkok Tower 4, the hub offers a modern environment designed to support high-quality clinical training. Purpose-built as a regional center of excellence, the Academy will upskill licensed aesthetic practitioners through science-based education, hands-on technique, and expert
- 김갑성 기자
- 2026-01-14 08:15
-

DNV SUPPORTS NOUL IN IVDR CERTIFICATION FOR AI-BASED MEDICAL DIAGNOSTIC PRODUCTS
SEOUL, South Korea, Jan. 14, 2026 -- DNV, the independent global assurance and risk management provider, is partnering with Korean AI-driven blood and cancer diagnostics company Noul to certify their malaria diagnosis, blood cell morphology and cervical cancer testing solutions, under the European Union's In Vitro Diagnostic Medical Device Regulation (IVDR). The EU's IVDR provides a regulatory basis for establishing the safety and effectiveness of new in-vitro diagnostic (IVD) devices. Most IVD devices require a certificate of compliance with the IVDR to receive a CE
- 김갑성 기자
- 2026-01-14 08:00
-

DNV, 노을(Noul)의 AI 기반 체외진단의료기기 대한 EU IVDR 인증 심사 진행
서울, 대한민국 2026년 1월 14일 -- 글로벌 인증기관(Notified Body, NB)인 DNV가 AI 기반 혈액 및 암 진단 플랫폼 기업 '노을(Noul)'의 주요 제품군에 대해 유럽연합(EU)의 체외진단의료기기 규정(IVDR)에 따른 인증 심사를 진행한다고 밝혔다. 이번 심사 대상은 노을의 혁신적인 miLab™ 플랫폼을 기반으로 하는 말라리아 진단용 miLab™ MAL 카트리지, 혈구 형태 검사용 miLab™ BCM 카트리지, 자궁경부암 검사용 miLab™ CER 카트리지 등 총 세 가지 솔루션이다. 노을의 miLab™ 플랫폼을 기반으로 하는 이 솔루션들은 통합 시료 전처리부터 디지털 이미징, AI 분석 기능을 단일 기기에 결합한 것이 특징이다. 이러한 기술적 결합을 통해 확보한 뛰어난 휴대성은 도심의 최첨단 병원뿐만 아니라 의료 인프라가 제대로 구비되지 않은 아프리카 등 오지의 시골 진료소에 이르기까지, 장소에 구애받지 않고 전 세계 모든 의료 환경에서 일관된 고품질 검사와 효율적인 워크플로우를 가능하게 한다. EU의 IVDR은 체외진단의료기기(IVD)의 안전성과 유효성을 철저히 검증하기 위
- 김갑성 기자
- 2026-01-14 08:00
-

Rakuten Medical and LOTTE Biologics Sign Manufacturing Agreement to Support Biopharmaceuticals in Global Oncology Program
SEOUL, South Korea, Jan. 14, 2026 -- Rakuten Medical, Inc. and LOTTE Biologics today announced that they have signed a biopharmaceutical contract manufacturing agreement during the J.P. Morgan Healthcare Conference in San Francisco to strengthen Rakuten Medical's production capabilities for its innovative oncology therapy, Alluminox® platform-based photoimmunotherapy. Under the agreement, LOTTE Biologics will provide advanced manufacturing services for monoclonal antibody intermediates and their conjugates, supporting Rakuten Medical's global clinical development an
- 김갑성 기자
- 2026-01-14 07:34
-

Firstsource Acquires TeleMedik To Bolster Digital Offerings and Expand Presence in U.S. Healthcare Payer and Provider Markets
MUMBAI, India and DALLAS, Jan. 14, 2026 -- Firstsource Solutions Limited (NSE: FSL) (BSE: 532809), a leading global provider of business management services and an RP-Sanjiv Goenka Group company, today announced the acquisition of TeleMedik, a Puerto Rico based pioneer in the development and implementation of technological solutions. The acquisition significantly strengthens Firstsource's end-to-end clinical and utilization management capabilities, expands its footprint across payer-provider networks, and further enriches the company's fully integrated and differentiated Business Proc
- 김갑성 기자
- 2026-01-14 03:19
-

ArisGlobal Accelerates Growth and AI Adoption Following Strong 2025 Performance
More than 100 new and expanded LifeSphere customers, 34 global go-lives, and rapid enterprise adoption of NavaX signal strong momentum BOSTON, Jan. 14, 2026 -- ArisGlobal, an AI-first technology company at the forefront of life sciences and creator of LifeSphere® , today announced strong results for 2025, marked by sustained customer growth, successful global implementations, and accelerated adoption of its NavaX AI-powered capabilities. Throughout the year, ArisGlobal helped pharmaceutical and life sciences organizations modernize Safety, Regulatory, and R&D IT operati
- 김갑성 기자
- 2026-01-13 23:54
-

Picarro Sets New Standard in Fenceline Monitoring for Hazardous Air Pollutants
Comprehensive Solution Delivers Real-Time, Defensible Measurements, Transforming Fenceline Monitoring into a Proactive Compliance and Risk Management Tool SANTA CLARA, Calif., Jan. 13, 2026 -- Picarro, Inc., a global leader in emissions monitoring, analytics, and services, today announced a breakthrough in air-quality compliance with the release of its Fenceline Solution. Picarro's Fenceline Solution features continuous fenceline monitoring, mobile monitoring, and cloud-based analytics working together in real-time. The new solution is designed to meet US EPA requirements for a
- 김갑성 기자
- 2026-01-13 23:12
-
Ractigen Therapeutics Announces First Patient Dosed in Phase II Clinical Trial of RAG-17 for SOD1-ALS
NANTONG, China, Jan. 13, 2026 -- Ractigen Therapeutics is pleased to announce that the first patient has been dosed in the Phase II clinical trial of RAG-17, an innovative siRNA therapy targeting SOD1-mutated amyotrophic lateral sclerosis (ALS). The initial dosing occurred at Second Affiliated Hospital, Zhejiang University School of Medicine, under the leadership of Dr. Zhi-Ying Wu, head of the Department of Medical Genetics/Center for Rare Diseases. This Phase II trial is a randomized, double-blind, placebo-controlled, multiple ascending dose (MAD) study designed to evaluate the safet
- 김갑성 기자
- 2026-01-13 20:30
-

Leo International Group Relocates Global Headquarters to Singapore Ahead of Its Centennial
Embodying a New Model of Inter-generational Enterprise Anchored in Healthcare, Finance and Education SINGAPORE, Jan. 13, 2026 -- As Leo International Group approaches its centennial year in 2026, the Group is not merely commemorating a hundred years of legacy and commercial success. With the formal relocation of its global headquarters to Singapore and the planned inauguration of its Family Office, Leo International Group is using this historic milestone to articulate its next century design; a renewed vision for what a modern enterprise must stand for in a rapidly changing wor
- 황정호 기자
- 2026-01-13 20:20
-
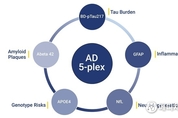
알라마 바이오사이언스, NULISAqpcr™ AD 5-플렉스 어세이 출시 발표...알츠하이머병 연구 시 혈액 기반 바이오마커 검출 진일보
[ 메디채널 김갑성 기자 ] 프레몬트, 캘리포니아주, 2026년 1월 13일 -- 질병 조기 발견 기술의 발전에 주력하는 정밀 단백질체학 선도 기업 알라마 바이오사이언스(Alamar Biosciences, 이하 "알라마")가 연구 전용(RUO) NULISAqpcr™ AD 5-플렉스 어세이 출시를 발표했다. 이는 알츠하이머협회(Alzheimer's Association) 권장 가이드라인과 관련된 5대 혈액 기반 바이오마커, 즉 뇌 유래 인산화 타우 217(BD-pTau217), 신경섬유 경쇄(NfL), 아밀로이드 베타 42(Aβ42), 아교섬유산성 단백질(GFAP), APOE4(보유자 상태)를 혈액 또는 혈장 샘플 하나로 정량적으로 동시 측정할 수 있는 혁신적인 솔루션이다. NULISAqpcr AD 5-플렉스 어세이는 주요 바이오마커를 멀티플렉스 포맷 하나로 통합하여 감도, 특이성, 사용 편의성이 탁월하다. 이 어세이는 BD-pTau217, Aβ42, NfL, GFAP를 통합하여 아밀로이드 및 타우 병리, 신경 퇴화 및 염증을 면밀히 모니터링할 수 있으며, 동시에 알츠하이머병의 유전적 위험 요인 중 가장 강력
- 김갑성 기자
- 2026-01-13 18:36
- 1포리프 굿마팻 (누바디 굿바이 마이팻) 다이어트 건강기능식품 출시
- 22021년 식품분석전문가 1급/2급 검정자격시험 안내
- 3투썸플레이스, 초콜릿 애프리콧 무스 출시
- 4하만카돈, AURA STUDIO 4 블루투스 스피커 출시
- 5아임웹, CRM 기능 출시… 고객 행동 데이터 기반 마케팅 지원
- 6대구오페라하우스 - 20주년 기념 골든 보이스 시리즈Ⅲ, 테너 콘서트
- 7만성질환자들의 유료독감백신접종에 정책적인 배려가 필요하다
- 82021년 대학생기자단 모집
- 9대한약물영양의학회 2020년 추계학술대회
- 10대한영양제처방학회 춘계학술대회 성황리에 마쳐
2026-03-03_TUE
- US BioTek Laboratories Completes Merger With NutriPATH Pathology 02:15
- Skyhawk Announces Australia's Therapeutic Goods Administration Has Determined SKY-0515 for Huntington's Disease Meets Eligibility Criteria for Registration via the Provisional Approval Pathway 01:20
- Innovaccer Receives Frost & Sullivan's 2026 United States New Product Innovation Recognition for Excellence in AI-Driven Patient Access Solutions 00:57
2026-03-02_MON
- NYSE Content Update: Georgia Gov. Kemp + Atlanta Mayor Dickens to Ring Bell Ahead of World Cup 23:21
- TDK opens its fifth regional headquarters in Asia-Pacific with a new business entity in India 23:00
- The Sigourney Award-2026 Now Accepting Applications for Psychoanalytic Innovation 22:49
- XellSmart Receives Fourth Consecutive US FDA & China NMPA IND Clearance for MSA-P iPSC-derived Cell Therapy 22:10










